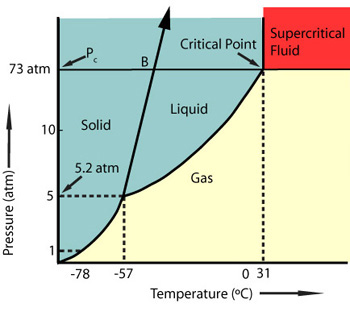
A supercritical fluid (SCF) is any substance above its critical temperature and pressure; the fluid has the properties of both gas and liquid while not exhibiting a distinct liquid or gaseous phase. Thus, an SCF is,
• Dense like a liquid to dissolve materials
• Has low viscosity, high diffusivity, no surface tension like a gas
Supercritical Fluids:
The Technology
Carbon dioxide is the most commonly used supercritical fluid and becomes supercritical at 31oC and 74 BAR. There are many applications for scCO2 outside the textile dyeing industry, most commonly used in decaffeination of coffee and extraction of natural herbs and spices.
Carbon dioxide is environmentally friendly, non-toxic and in our case most of the CO2 is internally recycled with very little vented to the atmosphere. scCO2 based textile operations represent a very “green” technology.
Carbon dioxide as a solvent is well suited for use in textile operations such as scouring, dyeing and post-treatments because it can be used a solvent whose density (hence solvent properties) can be modified by simply changing the temperature and pressure of the carbon dioxide. Carbon dioxide above its supercritical condition has the following attributes:
• scCO2 is a solvent that can dissolve dyes, oils,
surfactants, waxes etc.
• Solvent properties are tunable
• Behaves mostly like non-polar solvent e.g.
alkanes (low dielectric constant and dipole
moment)
• Has some quadrupolar characteristics; behaves
like dioxane
• scCO2 has some “Polar” attributes: Capable of
weak Lewis acid and base interactions;
Hydrogen bonding e.g. with amines;
Fluorocarbon interactions
• Low cost
• Safe, environmentally benign/non-toxic,
• Readily recyclable
• Does not oxidize
• Pressure reduction converts to gas hence
removal of scCO2 as solvent does not require
scCO2 for Dyeing
• Conventional drying required to remove solvents
such as water